 /accurate-illustration-of-the-periodic-table-82020791-57cc76f23df78c71b66efbd7.jpg)
However, IUPAC recommends that the numbers 1 through 18 be used, and these labels are more common. /periodic-table-of-elements-680789917-58ea3e903df78c5162f92b6f.jpg)
In the United States, the labels traditionally were numerals with capital letters. Groups are labeled at the top of each column. The elements are arranged in seven horizontal rows, called periods or series, and 18 vertical columns, called groups. Each box represents an element and contains its atomic number, symbol, average atomic mass, and (sometimes) name. A modern periodic table arranges the elements in increasing order of their atomic numbers and groups atoms with similar properties in the same vertical column. The modern statement of this relationship, the periodic law, is as follows: the properties of the elements are periodic functions of their atomic numbers. (credit a: modification of work by Serge Lachinov credit b: modification of work by “Den fjättrade ankan”/Wikimedia Commons)īy the 20th century, it became apparent that the periodic relationship involved atomic numbers rather than atomic masses. (a) Dimitri Mendeleev is widely credited with creating (b) the first periodic table of the elements. Although Mendeleev and Meyer had a long dispute over priority, Mendeleev’s contributions to the development of the periodic table are now more widely recognized. The discoveries of gallium (1875) and germanium (1886) provided great support for Mendeleev’s work. But Mendeleev went one step further than Meyer: he used his table to predict the existence of elements that would have the properties similar to aluminum and silicon but were yet unknown. Both published tables with the elements arranged according to increasing atomic mass. Fluorine (F), chlorine (Cl), bromine (Br), and iodine (I) also exhibit similar properties to each other, but these properties are drastically different from those of any of the elements above.ĭmitri Mendeleev in Russia (1869) and Lothar Meyer in Germany (1870) independently recognized that there was a periodic relationship among the properties of the elements known at that time. For example: Li, Na, and K are much more reactive than are Ca, Sr, and Ba Li, Na, and K form compounds with oxygen in a ratio of two of their atoms to one oxygen atom, whereas Ca, Sr, and Ba form compounds with one of their atoms to one oxygen atom. However, the specific properties of these two groupings are notably different from each other. A second grouping includes calcium (Ca), strontium (Sr), and barium (Ba), which also are shiny, good conductors of heat and electricity, and have chemical properties in common. One such grouping includes lithium (Li), sodium (Na), and potassium (K): these elements all are shiny, conduct heat and electricity well, and have similar chemical properties. Identify metals, nonmetals, and metalloids by their properties and/or location on the periodic table.Īs early chemists worked to purify ores and discovered more elements, they realized that various elements could be grouped together by their similar chemical behaviors.Predict the general properties of elements based on their location within the periodic table.State the periodic law and explain the organization of elements in the periodic table.Not to miss out, element 102, Nobelium, No, is named in honor of Alfred Nobel, who set aside his vast fortune to establish Nobel Prizes. Mendeleev never received a Nobel Prize for his work, but element 101 was named Mendelevium, Md, after him. For instance they discovered phosphorus when they isolated it from urine. Over time these gaps have gradually been filled in as scientists unearthed new elements. Not only did Mendeleev arrange the elements in the correct way, but he also had the foresight to leave gaps for undiscovered elements. At that time, he had only 50 elements to arrange. 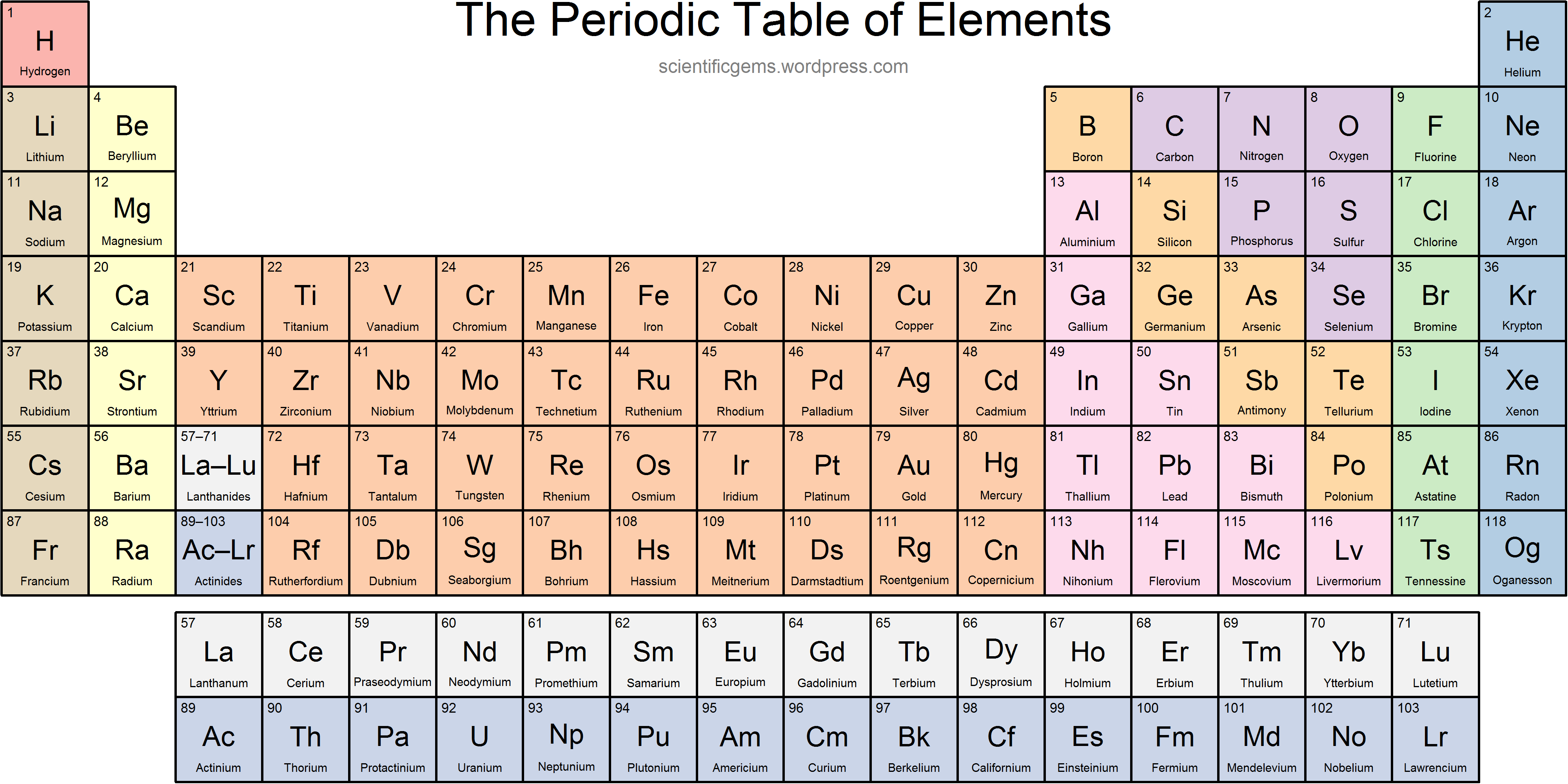
He wrote the properties of the elements on pieces of card and rearranged them until he realised that, by putting them in order of increasing atomic weight, certain properties of elements regularly occurred. Then in 1869, a Russian scientist called Dmitri Mendeleev produced one of the first practical periodic tables. Several other attempts were made to group elements together over the coming decades. The earliest attempt to classify the elements was in 1789, when Antoine Lavoisier grouped the elements based on their properties into gases, non-metals, metals and earths. The discovery of other elements followed regularly and soon it became necessary to arrange them in some sort of order. However, the first scientific discovery of an element occurred in 1649 when Hennig Brand discovered phosphorous. Copper has been used by humans for as much as 7000 years and elements such as gold, silver, tin, lead and mercury have been known for many thousands of years. 
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
